Systematic literature review consulting and software
Our experts combine experience with technology to streamline the end-to-end literature review process.
Systematic literature reviews can be herculean undertakings. That’s why we built Litstream. This tool allows researchers to collaborate on literature screening and data extraction through an efficient platform that tracks the literature process meticulously and across multiple team members working simultaneously. Managers can design fit-for-purpose review steps, make assignments, and monitor progress at a granular level. Litstream helps ensure transparent documentation of the review process while prioritizing the most relevant literature to your team of subject matter experts.

End-to-end support, from experts who have seen it all
We don’t just offer a product—we offer a partnership. Our team of systematic literature review experts is tailor-made to help you navigate the comprehensive review process, from start to finish. We have decades of hands-on experience across research spheres, developing search strategies, establishing inclusion criteria, assessing study quality, supporting data integration and data visualization, performing risk analyses, and helping make policy recommendations for regulators.

Making literature review efficient and painless
Whether you want to thematically isolate existing trends or sift through mountains of evidence to foreground research questions, Litstream makes achieving your goals fast and painless. Our literature review tool automates the flow of studies from one step to the next in your review process to ensure that the record of decision is clearly traceable. We help our clients develop interoperability amongst data systems by building automated pipelines that migrate data seamlessly from platform to platform.

One-of-a-kind understanding of the literature review process
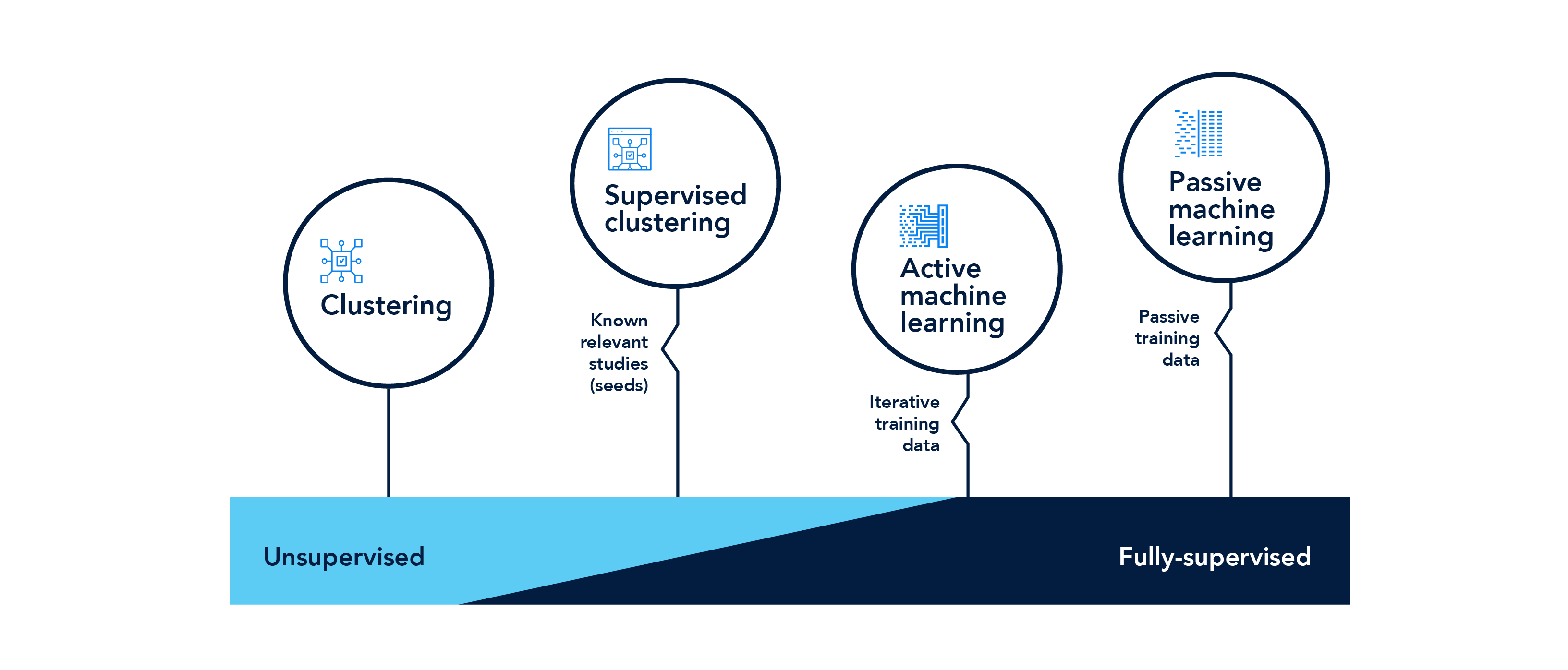
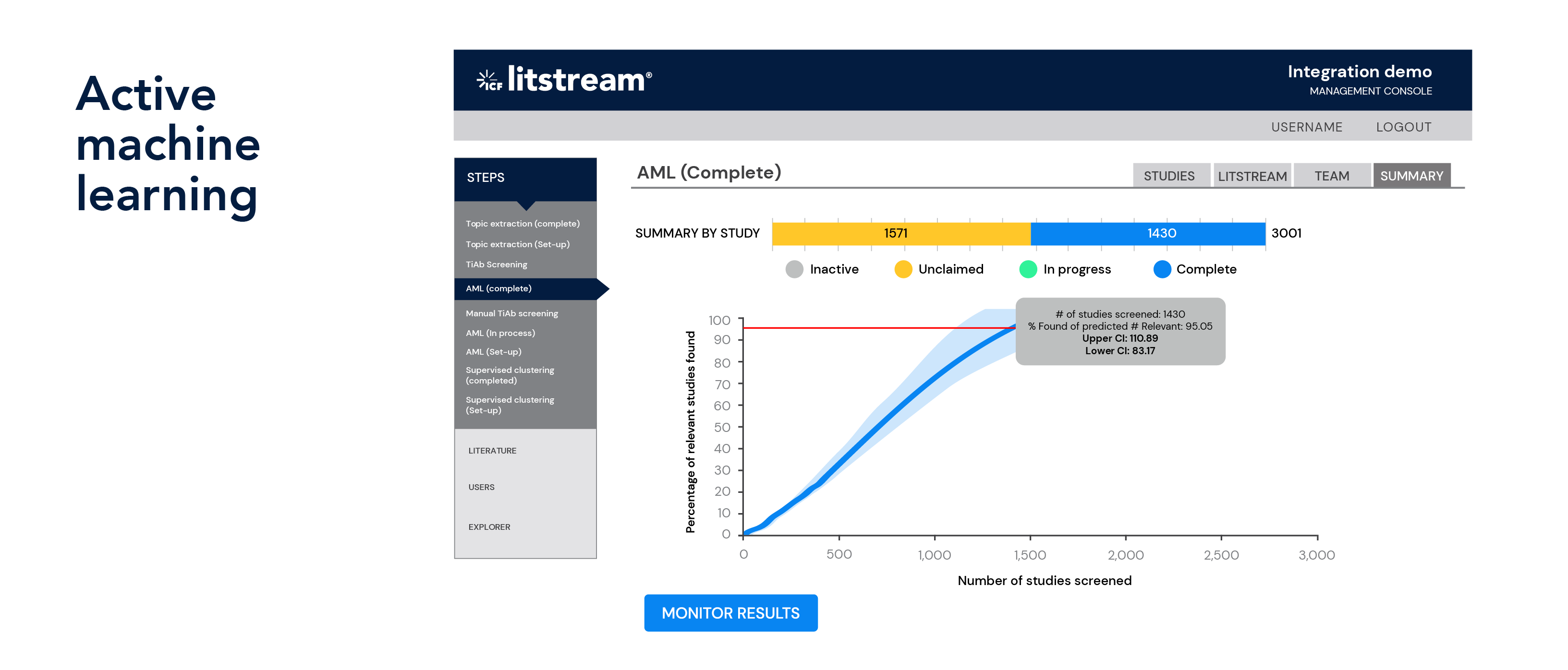
We offer a toolbox of four prioritization approaches (topic extraction, supervised clustering, machine learning and active machine learning) to help you find the literature you should focus on while filtering out the noise or off-topic literature that can consume valuable time. In a simulated study, we estimated that over 1,000 project hours could have been saved by using our built- in supervised clustering approach to prioritize screening efforts across six EPA Integrated Risk Information System risk assessments. Our staff is well versed in the application of our prioritization approaches, working with clients to use the best approaches in each situation to deliver results with the least amount of bias. Since we designed these tools while doing the work, our team has a one-of-a-kind understanding of the ins and outs of the literature review process.
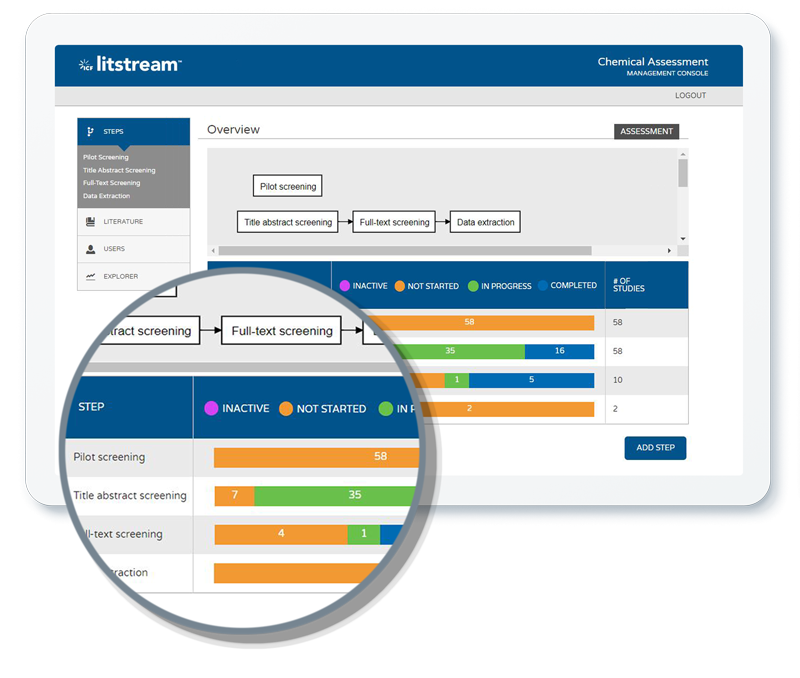
Complete transparency, unparalleled accuracy
When it comes to systematic literature review, transparency is paramount. Organizations should follow a process that is exhaustively documented and easily reproducible. Work must hold up to public scrutiny and stringent outside review. Beyond that, our fully operationalized machine learning tools have outcompeted costly and time-intensive custom algorithms in study after independent study. From documenting the literature search strategy, to describing prioritization methods, and following each study through the systematic review steps, Litstream is a holistic solution to manage your studies and extracted data with reporting and visualization features to help you build all the products you need to support your assessment. Our team of expert practitioners can help you utilize systematic review tools effectively and integrate them with your organizational resources to create a unique solution that is tailored to your needs.
Litstream’s literature review tool suite
Litstream’s tool suite covers literature search, prioritization, text screening, inventory, data extraction and analysis, risk-of-bias or study quality assessments, and visualization. These tools (1) eliminate duplicates and redundancies early on in the process, freeing up attention and human capital; (2) narrow the body of literature that will be reviewed in the downstream parts of the systematic review process; and (3) prioritize literature that best answers the research questions such that subject matter experts can dive into the evidence and speed up the rate of discovery.
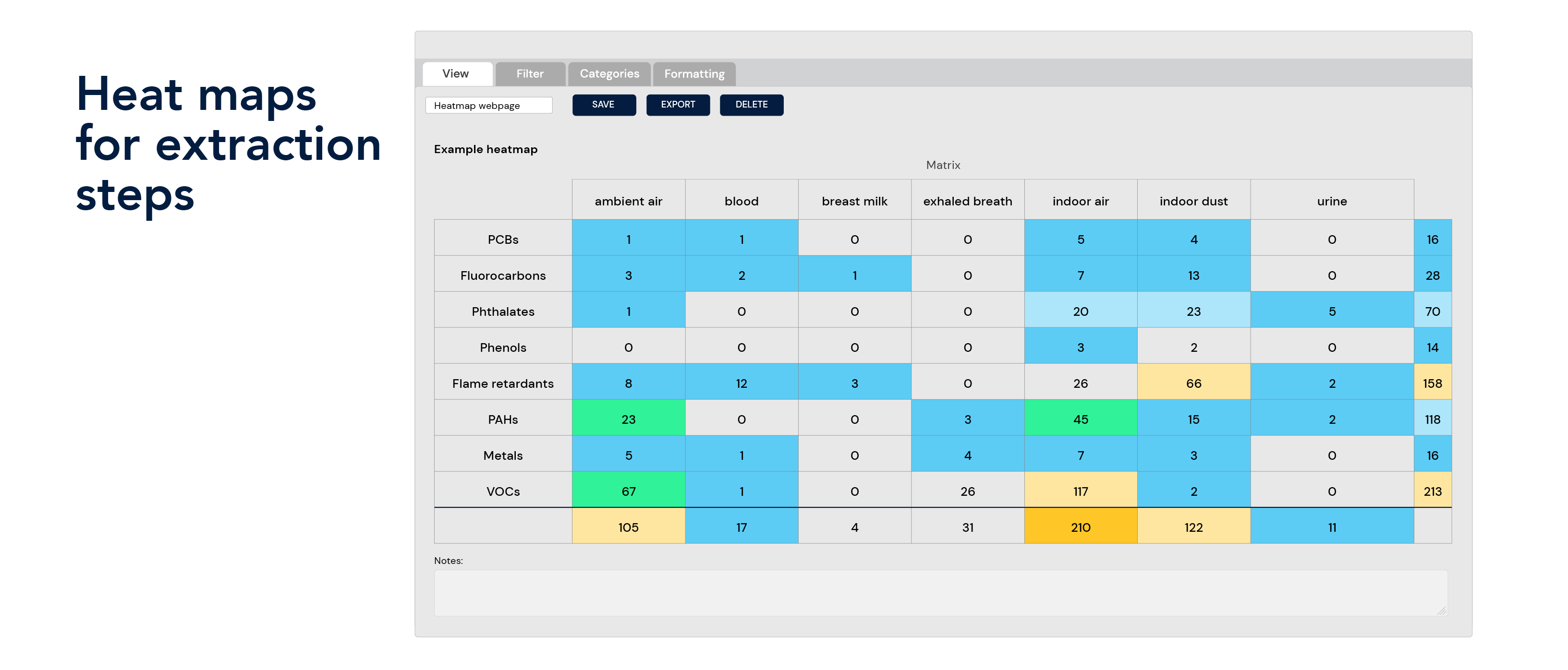
Flexible data extraction
Document tagging
Design document tagging categories and keyword highlights to be used in the title or abstract and full-texting screening steps. Assign screening tasks based on the availability and expertise of screeners. Perform multiple levels of review and move studies into downstream steps based on final screening results.
Keyword analysis and document clustering
Cluster studies by common words or text similarities. Users can create keyword hierarchies to review document clusters with the most relevant keywords or use seed references to identify clusters containing high proportions of relevant literature to target manual screening efforts.
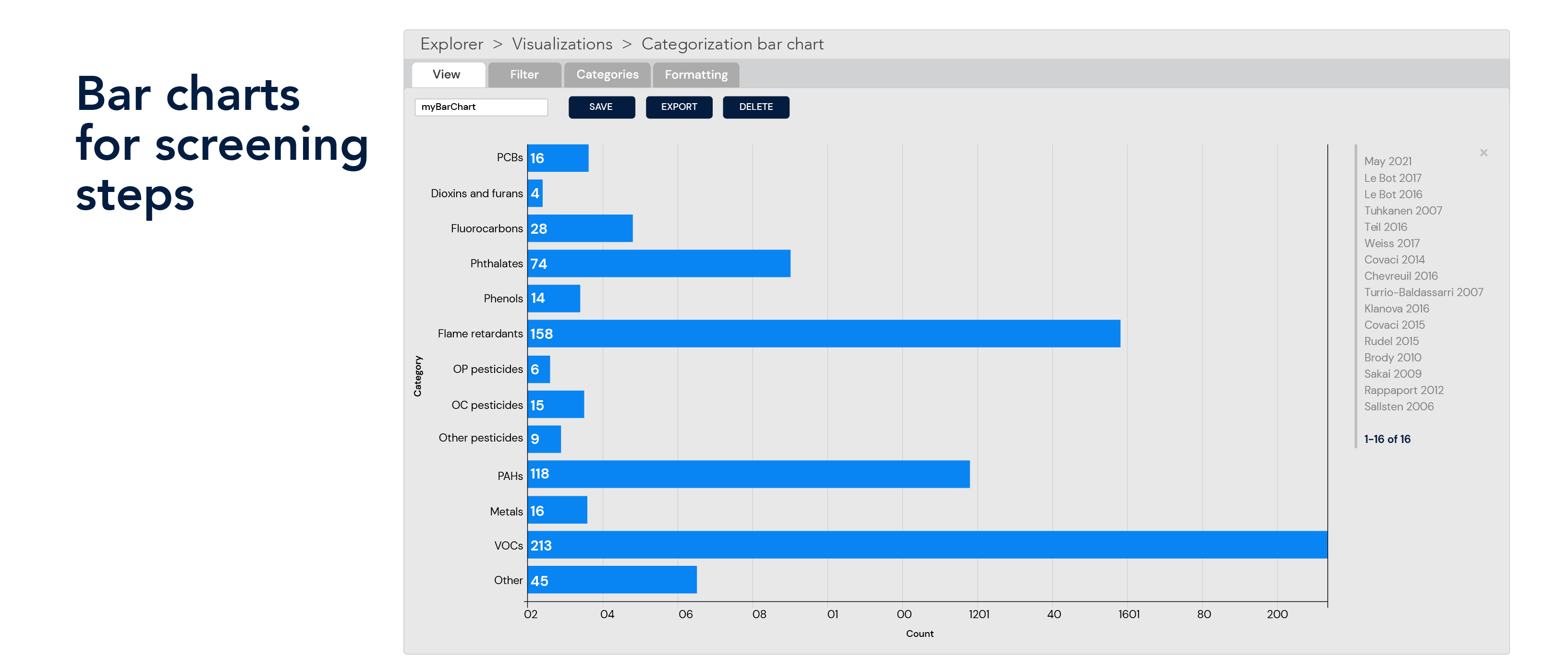
Data reporting and visualization
Document prioritization and multi-track approach
Prioritize documents for review using multiple approaches, including active machine learning, machine learning, unsupervised clustering, and supervised clustering. Analyze titles and abstracts to find the most relevant studies and reduce the amount of time spent reviewing off-topic literature.
In-app collaboration and workflow
A diverse range of applications
Our literature review tools have been used to great effect by state and federal agencies, nonprofits, watchdogs and private researchers. Over successive decades we have supported major aviation and transportation projects, large-scale psychological and environmental impact surveys, and exhaustive scientific and medical analyses, always with an eye toward versatility and interoperability. We ensure that, regardless of scale, our literature review tool is exactly what the project requires.
Our services
Searching and text analytics expertise
- Automated data collection and reference deduplication
- Sorting literature based on relevance
- Building seed sets and guided machine learning
- Streamlined literature characterization based on keywords and complex heuristics
- Automated full text highlighting using keywords and named entity recognition approaches
Problem formulation and end-to-end collaboration
- Collaborative problem formulation based on clients and stakeholders
- Training support for collaborators
- Decades of hands-on systematic review expertise
- End-to-end support ensuring ease of product use, and meeting project goals
Related industries, services, and solutions